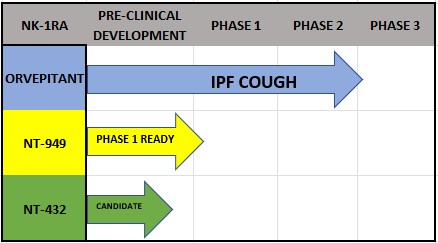
PIPELINE
ORVEPITANT:
- A potent and selective antagonist of the human neurokinin-1 (NK1) receptor.
- Has been evaluated in more than 900 healthy subjects and patients and has been shown to be well tolerated with a good safety profile.
- Pharmacokinetic profile allows once daily dosing with a simple oral tablet formulation.
- Fully occupies brain neurokinin-1 receptors throughout a 24-hour dose interval at the preferred clinical dose.
- In Phase 2 clinical development as a treatment for chronic cough disorders associated with hypersensitivity of the cough reflex pathway.
- Orvepitant is currently being evaluated in chronic cough associated with IPF. A phase 2 randomised, double-blind, placebo-controlled 2 period cross-over study is currently ongoing (clinicaltrials.gov ID NCT05185089).
FOCUS – Chronic Cough in Idiopathic Pulmonary Fibrosis (IPF)
Idiopathic pulmonary fibrosis (IPF) is a chronic respiratory condition, occurring primarily in older adults, that is characterised by the progressive formation of irreversible scar tissue in the lungs.
IPF is a rare disease that has been designated as an orphan condition [1]. The prognosis for IPF is worse than that of many cancers and other forms of lung fibrosis, with median life expectancy after diagnosis of just 2 to 5 years [2]. The cough associated with IPF can be debilitating in these terminally ill patients and may contribute to disease progression.
Cough is a common symptom in patients with IPF that is often severe and difficult to treat [3] and is frequently cited as one of the most burdensome aspects of the disease [4]. Chronic cough, regardless of its cause, can impact markedly on health-related quality of life [5]. Co-morbid sequelae of repeated coughing can include musculoskeletal chest pain, fatigue, sleep disturbance, urinary incontinence and, in extreme cases, syncope or rib fractures.
IPF cough is also an independent marker of disease [6]. Cough in IPF may not just be a symptom but could, by inducing repeated tractional injury, augment the activation of profibrotic mechanisms and therefore promote disease worsening [7].
There is hypersensitivity of the cough reflex pathway in IPF
While the pathophysiology of cough in IPF is not yet fully understood, data points to the development of hypersensitivity in the cough reflex pathway [8]. Possible explanations for the hypersensitivity are that mechanical distortion of the lung, caused by the fibrosis, directly influences afferent nerve fibres, and/or that efferent nerves that inhibit cough are destroyed by the fibrosis.
Clear evidence that IPF patients have an increased cough reflex sensitivity is provided by studies showing an amplified response to inhaled capsaicin compared to healthy controls [9, 10] and to mechanical stimulation of the chest wall [11]. Furthermore, inhalation of Substance P induced cough in patients with IPF (but not in healthy controls) suggesting up-regulation of pulmonary afferent C-fibres [10].
Orvepitant has been shown to reduce cough burden in cough hypersensitivity syndrome
Orvepitant is a selective antagonist of the human neurokinin-1 receptor which blocks the effects of its cognate agonist, Substance P. Clinical proof of concept in cough hypersensitivity syndrome has been established with orvepitant in the VOLCANO-2 chronic cough study. In this study, clinically relevant and statistically significant improvements were shown on several patient reported assessments of cough burden. Patients with cough due to IPF also have cough hypersensitivity syndrome and so they can be expected to experience the same beneficial outcomes with orvepitant therapy.
- Orphanet ORPHA:2032
- Meltzer EB & Noble PW, 2008
- Key AL et al, 2010
- https://www.fda.gov/media/91396/download
- Morice et al, 2007
- Ryerson CJ et al, 2011
- Sato R et al, 2019
- van Manen MJG et al, 2016
- Doherty MJ et al, 2000
- Hope-Gill B et al, 2003
- Jones RM et al, 2011

